PeeBee wrote: ↑Wed Jan 29, 2020 11:24 am
… There's so little in your water you can pretty well ignore "alkalinity", …
I've got away with making a statement like that, so far. So to flesh it out a bit and its a good excuse to post a colourful map (ooo; I do like them):
It's okay for me and you ("Brown Beer") to ignore alkalinity, but that might be taken as an inflammatory statement (I have a gift for annoying people too). "Alkalinity" (and "residual alkalinity") is a valuable tool for calculating what needs to be added to water before making additional "tweaks" to get the mash in the right pH range. But for our water we're already skipping straight to the "tweak" stage. "Alkalinity" has two objectives: Firstly to help predict and adjust the outcome of a mash for those that need it, and secondly, to pulp the brains of those that don't need it.
And to see who does and doesn't need it (time to get a colourful map out):
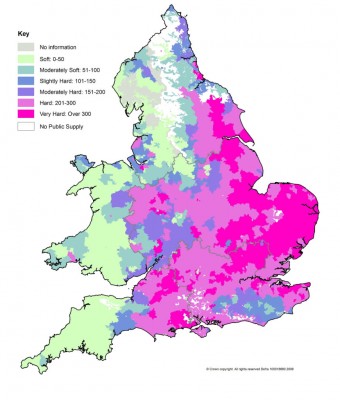
From the map you can judge that we really are in the minority (of UK population). It doesn't make the difference between "temporary hardness" (or "carbonate hardness" - primarily responsible for "alkalinity") and "permanent hardness" (calcium and magnesium salts), but it generally follows in the UK that the areas are the same (a figure I saw was 60% of UK users have CaCO3 levels greater than 200ppm). And the fine tweaks can be left up to the numerous calculators. The calculators are not all the same when it comes to pH predicting, I used to use Bru'n Water but its predictions no longer work for me, so I'm trying "Mash Made Easy" which has the added advantage of being able to "tweak" how it predicts pH outcome (which promptly has it leave the "Easy" arena).
<EDIT: Something I whinge about, and here I am doing it myself! "60% of UK users have
<hardness as> CaCO3 levels greater than 200ppm".
As daft
as I am, or should that be "a daft ass I am".>.
. I guess I don't need Camden tablet addition? I've always just added it...


 . I guess I don't need Camden tablet addition? I've always just added it...
. I guess I don't need Camden tablet addition? I've always just added it...
