AMS consistency
-
Matt in Birdham
- Drunk as a Skunk
- Posts: 764
- Joined: Thu Oct 08, 2015 10:27 pm
AMS consistency
I missed mash pH to the upside for my last 3 brews - consistently, hitting 5.6 instead of a targeted 5.4 (ended up correcting with lactic on all occasions, no harm done to the beer). All times I have been using AMS plus a little gypsum and CaCl.
Tonight I finally decided to test my AMS and try and work out what is going on, and after adding 5ml of AMS to 5l of water the alkalinity dropped from 249 to 108. Calculators suggest that this should have be to ~60 ppm CaCO3. The interesting thing is that this has happened over the course of two bottles - finishing up one, and starting a new one last brew (and test carried out with the new one as well).
Any thoughts? Is this common? Am I screwing something up? I've tried running through two calculators (Graham's + Brewersfriend) and also using the numbers directly from the malt miller.
Tonight I finally decided to test my AMS and try and work out what is going on, and after adding 5ml of AMS to 5l of water the alkalinity dropped from 249 to 108. Calculators suggest that this should have be to ~60 ppm CaCO3. The interesting thing is that this has happened over the course of two bottles - finishing up one, and starting a new one last brew (and test carried out with the new one as well).
Any thoughts? Is this common? Am I screwing something up? I've tried running through two calculators (Graham's + Brewersfriend) and also using the numbers directly from the malt miller.
- Dennis King
- Telling everyone Your My Best Mate
- Posts: 4229
- Joined: Sat Aug 11, 2007 7:52 pm
- Location: Pitsea Essex
Re: AMS consistency
AMS comes from Murphy's. Some of their products/tests have been know to fluctuate.
- Eric
- Even further under the Table
- Posts: 2920
- Joined: Fri Mar 13, 2009 1:18 am
- Location: Sunderland.
Re: AMS consistency
Murphy's advise that 35ml of AMS per hl will reduce alkalinity by 64mg/l, that is 183mg of alkalinity per ml of AMS. Their specification has a tolerance of + of - 5%, so 174 to 192 while your findings are significantly outside that range.
While I've had problems with Murphy's products and services, AMS has always been to specification, although it is maybe 4 years since last buying AMS.
It is advisable to check alkalinity after acid treatment of brewing liquor, but maybe some calculators don't make allowance for such precautions.
While I've had problems with Murphy's products and services, AMS has always been to specification, although it is maybe 4 years since last buying AMS.
It is advisable to check alkalinity after acid treatment of brewing liquor, but maybe some calculators don't make allowance for such precautions.
Without patience, life becomes difficult and the sooner it's finished, the better.
-
Matt in Birdham
- Drunk as a Skunk
- Posts: 764
- Joined: Thu Oct 08, 2015 10:27 pm
Re: AMS consistency
Thanks. Yeah, it's a little weird since it does seem a long way off, but no reason to believe my alkalinity measurements are wrong (have tried with 2 kits). I'll adjust my next batch in stages, checking the alkalinity drop with AMS before adding any other minerals and see if that gets me there. Annoying, though, if it is the AMS but if others aren't experiencing this then I still wonder if something else is going on..
- Eric
- Even further under the Table
- Posts: 2920
- Joined: Fri Mar 13, 2009 1:18 am
- Location: Sunderland.
Re: AMS consistency
It's just as likely that a great many just use the amount given by the calculator and don't check the result. In many cases it won't matter significantly but for those with variable or very high alkalinity making pale beers using acids from unreliable sources are bound to have inconsistent results which can lead to misleading conclusions.Matt in Birdham wrote:Thanks. Yeah, it's a little weird since it does seem a long way off, but no reason to believe my alkalinity measurements are wrong (have tried with 2 kits). I'll adjust my next batch in stages, checking the alkalinity drop with AMS before adding any other minerals and see if that gets me there. Annoying, though, if it is the AMS but if others aren't experiencing this then I still wonder if something else is going on..
The only concern I'd have about that test is that sufficient time was allowed for the reactions to complete and liberate carbon dioxide. My preferred method is with a pH meter, measuring the volume of acid required in a suitable volume of water, to read pH 4.4 where I assume all alkalinity has been neutralised.
Without patience, life becomes difficult and the sooner it's finished, the better.
-
jaroporter
- Drunk as a Skunk
- Posts: 996
- Joined: Sat Jul 21, 2012 11:12 pm
- Location: Garden of England
Re: AMS consistency
this is something i always wonder about, but have never seen quantified (just that "time should be taken"). i suppose i could run a series of tests to see how quickly it's doing things, but it's just lazier to just procrastinate on JBK and leave the liquor a very conservative length of time! anyone know roughly how long "time" is, or is it going to be relative to amount of treatment needed? think i'll have to get the kit out soon..Eric wrote: The only concern I'd have about that test is that sufficient time was allowed for the reactions to complete and liberate carbon dioxide.
dazzled, doused in gin..
- Eric
- Even further under the Table
- Posts: 2920
- Joined: Fri Mar 13, 2009 1:18 am
- Location: Sunderland.
Re: AMS consistency
The period varies to a degree. The reaction between a small amount of acid into a volume of water with an excess of alkalinity will quickly complete, it would all happen in a small volume where the acid entered and it is necessary to mix the liquor well to be able to get a representative reading.
At the other end of the scale a large volume of acid capable of exactly neutralising all alkalinity present needs to be well stirred before the reaction will complete. An acid heavier than water can quickly sink to the bottom leaving unaffected water with virtually no alkalinity reduction on top of a dilute acid mix with no alkalinity.
Such processes can be observed using a pH meter immersed in alkaline water when slowly adding acid.
I have no potable water supply to where I brew and use 10 litre foodsafe buckets to carry tapwater @ 8 litre per bucket. Initial alkalinity is measured and the appropriate amount of the determined acid added to each bucket and stirred before being poured into the HLT. After weighing the required grains and giving the HLT a few stirs a sample is taken and measured when any necessary adjustment is made, which is normally addition of a fraction of a ml of acid or a part of a litre of tapwater. There is no reason to be standing around doing nothing on a brewing day.
At the other end of the scale a large volume of acid capable of exactly neutralising all alkalinity present needs to be well stirred before the reaction will complete. An acid heavier than water can quickly sink to the bottom leaving unaffected water with virtually no alkalinity reduction on top of a dilute acid mix with no alkalinity.
Such processes can be observed using a pH meter immersed in alkaline water when slowly adding acid.
I have no potable water supply to where I brew and use 10 litre foodsafe buckets to carry tapwater @ 8 litre per bucket. Initial alkalinity is measured and the appropriate amount of the determined acid added to each bucket and stirred before being poured into the HLT. After weighing the required grains and giving the HLT a few stirs a sample is taken and measured when any necessary adjustment is made, which is normally addition of a fraction of a ml of acid or a part of a litre of tapwater. There is no reason to be standing around doing nothing on a brewing day.
Without patience, life becomes difficult and the sooner it's finished, the better.
-
Matt in Birdham
- Drunk as a Skunk
- Posts: 764
- Joined: Thu Oct 08, 2015 10:27 pm
Re: AMS consistency
I didn't perhaps leave the water for as long as I should have (only 5-10 mins), although I did stir well. I do another batch tonight and see what happens.
- Wonkydonkey
- Drunk as a Skunk
- Posts: 847
- Joined: Sat Mar 01, 2014 9:37 am
- Location: In the Stables
Re: AMS consistency
I have two batches, 1 ams the other crs, yep I know thy are the same. But form two different peeps,
Anyhow I'm a slacker and grabbed the wrong bottle and added the usual amount to the water (without testing the water first) and then tested the water later with a saliferrtt kit and it was a bit more than off what I thought it should be, so I had to add a bit more tap water
So all I can say is test before and test after a few hrs. Or what ever time it should take at the temp of the reaction is in your bucket
Anyhow I'm a slacker and grabbed the wrong bottle and added the usual amount to the water (without testing the water first) and then tested the water later with a saliferrtt kit and it was a bit more than off what I thought it should be, so I had to add a bit more tap water
So all I can say is test before and test after a few hrs. Or what ever time it should take at the temp of the reaction is in your bucket
To Busy To Add,
-
BenB
Re: AMS consistency
I suspect I might be guilty of not waiting as well. In fact I'm pretty sure I don't usually- I just squirt it in from a syringe whilst the mash tun is filling from the HLT (which takes a couple of minutes).
- Kev888
- So far gone I'm on the way back again!
- Posts: 7701
- Joined: Fri Mar 19, 2010 6:22 pm
- Location: Derbyshire, UK
Re: AMS consistency
FWIW, AMS/CRS has generally been consistent for me in the past, though I haven't bought any very recently. It isn't impossible though, mistakes can and do happen with acid strengths (and Murphys haven't got a perfect record IIRC). Another possibility (which may be supported by it happening with different bottles of AMS) is that your measurement of alkalinity is out in some way - e.g. contaminated/dodgy test kit or some human error/misunderstanding.
As Eric mentioned, I'd guess many people just add the prescribed amount and the nearest they get to checking is via the mash pH. But mash pH is affected by other things, and often measured with pH papers that are vague at best, so it isn't really a very certain indicator that the acids were spot on.
Personally, I always treat the water after filling the HLT, and later test that both the alkalinity and calcium of the liquor are as they should be before using it. There have occasionally been mistakes (made by me) that this has caught before committing to the mash. But it wouldn't help if my tests results were wrong for some reason.
As Eric mentioned, I'd guess many people just add the prescribed amount and the nearest they get to checking is via the mash pH. But mash pH is affected by other things, and often measured with pH papers that are vague at best, so it isn't really a very certain indicator that the acids were spot on.
Personally, I always treat the water after filling the HLT, and later test that both the alkalinity and calcium of the liquor are as they should be before using it. There have occasionally been mistakes (made by me) that this has caught before committing to the mash. But it wouldn't help if my tests results were wrong for some reason.
Kev
- Eric
- Even further under the Table
- Posts: 2920
- Joined: Fri Mar 13, 2009 1:18 am
- Location: Sunderland.
Re: AMS consistency
Did you get chance to do a further test?Matt in Birdham wrote:I didn't perhaps leave the water for as long as I should have (only 5-10 mins), although I did stir well. I do another batch tonight and see what happens.
Thought I would check my AMS (bought direct from Murphy) and also observe how acid additions might differentially alter pH.
Such TDS meters as supplied display the amount in ppm of common salt in solution that would produce the same conductivity as the sample. Tapwaters contain other salts which influence conductivity by different amounts and therefore the amount displayed is unlikely to be valid. My water's TDS was determined to be the total of 9 major ions in WallyBrew's analyses.With saved water samples and those results it proved possible to recalibrate a meter to give sufficiently accurate readings.

My supply's mineral content varies, but mostly is near to its upper limit as in this case. It can half after periods of prolonged and heavy rain, presumably due to dilution. This makes it essential to measure alkalinity for every brew. One sample analised by WallyBrew had alkalinity of 255ppm CaCO3 with TDS of 541ppm. From this it may be assumed that with a TDS reading of 543, alkalinity would be the order of 256ppm as CaCO3. A Salifert kit showed alkalinity to be 5.13 meq/l, equivalent to 256.5ppm as CaCO2.
Obviously a large amount of luck happened here, but both pictures are genuine, surprising me as much as they may you and this mwthod has proved highly satisfactory with my water supply.
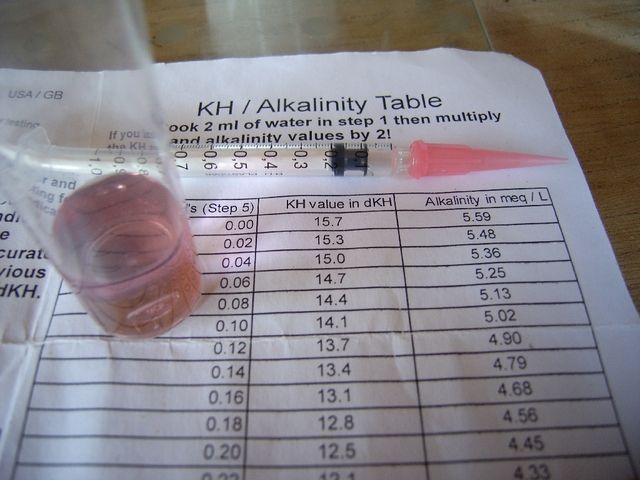
The pH meter was calibrated at 4.00 and 7.00 and half a litre of tapwater placed in a glass container with a 1ml syringe loaded with AMS.
The pH probe was placed in the water at one side of the vessel, resting on the bottom, while AMS was dropped at the opposite side in increments of 0.05ml. The meter reading was observed, allowing time for the reaction to complete, aided by stirring.
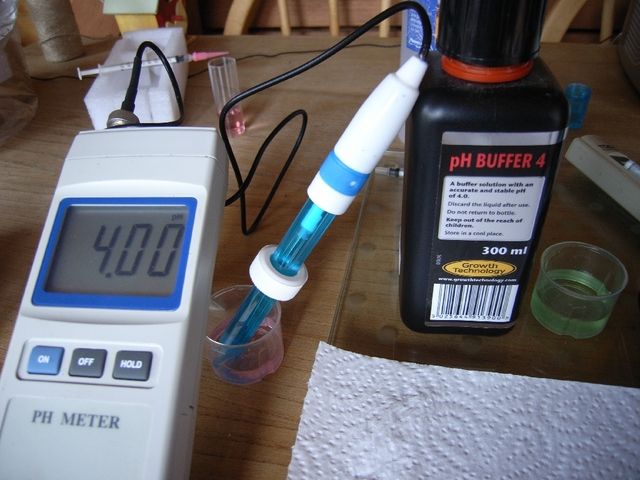
Initially pH was >7, requiring 1.5ml AMS for it to be less than 7. With the first drops the meter didn't alter until the liquid was stirred. Later pH would drop swiftly and significantly and stirring caused pH to rise again as the acid at the bottom near the probe was dispersed. When pH was around 6.35 the acid additions would temporarily reduce the meter reading, its value returning to almost what it was before the addition. From 0.5ml AMS pH started dropping quickly and after 0.65ml AMS pH settled at 5.58. A further 0.05ml AMS was more than enough to neutralise the remaining alkalinity reducing pH to below 4.5, the point recognised at which no alkalinity will exist.

From this it was found that 0.7ml of my AMS neutralised a little more than the total alkalinity present. Half a litre with alkalinity of 256mg/l would contain the equivalent of 128mg of CaCO3. This meant that 0.7ml of AMS had neutralised slightly more than 128mg of CaCO3 and 1ml would neutralise just over 183mg of CaCO3. (128/0.7)
Without patience, life becomes difficult and the sooner it's finished, the better.
Re: AMS consistency
Also in addition to the above. After you have worked out how much AMS/CRS to use, only add 75% of that amount, leave for 15 mins ensuring well mixed and stirred, and then test again and add required amount accordingly.
- scuppeteer
- Under the Table
- Posts: 1512
- Joined: Fri Oct 01, 2010 7:32 pm
- Location: Brenchley, Kent (Birthplace of Fuggles... or is it?)
Re: AMS consistency
Sorry for stating the obvious but do you check your raw liquor every time? It can vary somewhat. Also Murphys conduct their evaluation differently to others. I had a report back that stated the Raw Ph was 6.5, I measured it a 7.7. Something to do with how they measure the ions in the water to ascertain their results. Left me confused.
Dave Berry
Can't be arsed to keep changing this bit, so, drinking some beer and wanting to brew many more!
Sir, you are drunk! Yes madam, and you are ugly, but in the morning I shall be sober! - WSC
Can't be arsed to keep changing this bit, so, drinking some beer and wanting to brew many more!
Sir, you are drunk! Yes madam, and you are ugly, but in the morning I shall be sober! - WSC

